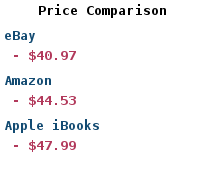Concurrent Programming on Windows

Book Description
Author Joe Duffy has risen to the challenge of explaining how to write software that takes full advantage of concurrency and hardware parallelism. In Concurrent Programming on Windows, he explains how to design, implement, and maintain large-scale concurrent programs, primarily using C# and C++ for Windows.Concurrent Programming on Windows has four major sections: The first introduces concurrency at a high level, followed by a section that focuses on the fundamental platform features, inner workings, and API details.
Book Details | |
| Publisher: | Addison-Wesley |
| By: | Joe Duffy |
| ISBN-13: | 9780321434821 |
| ISBN-10: | 032143482X |
| Year: | 2008 |
| Pages: | 1008 |
| Language: | English |
Book Preview | |
| Online | Concurrent Programming on Windows |
Paper Book | |
| Buy: | Concurrent Programming on Windows |